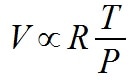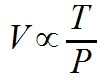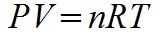WEEK 10
Boyle's and Charles's Laws--How Gases Behave
(SU ch. 11, LG ch. 3)
An Experiment on a Bird in the Air Pump, Joseph Wright, 1768, National Gallery, London, Public Domain PD-1923
DAY 1: Boyle's Law
READING
From "beginning of chapter" to "...the air to rush out again."
NARRATION PROMPTS
1. Tell what you know of Boyle's Law.
TIMELINE AND BOOK OF CENTURIES
NOTES
None yet.
|
DAY 2: A Cartesian Diver & Charles's Law
READING
From "Here's how to make..." to "...according to Charles's Law."
TIMELINE AND BOOK OF CENTURIES
NOTES
None yet.
|
DAY 3: The Ideal Gas Law
READING
From "In 1824 Gay-Lussac..." to "end of chapter"
NOTES
The equation for the Ideal Gas Law in this chapter is not correct. It would be correct if instead of being stated as an equality it was stated as a proportionality.
If the goal is to think about the relationship conceptually, because it is a constant, R can be dropped as well.
Here we can clearly see the simple relationships. For a constant pressure, if volume increases, so must temperature. For a constant temperature, if volume increases, the pressure must decrease. For a constant volume, if temperature increases, so must pressure.
There is a more common version of the ideal gas law. The n you see is what was left out of the equation in the book, and, without it, the equation is incorrect. There is quite a bit more which would need to be understood about this equation to be able to use it for solving problems. What is n? What is R? What are the units of all these variables? A more advanced physics or chemistry book will cover that information. The important thing to take away from this chapter is the relationship between V, T, and P.
|